CDC Approves COVID-19 Vaccination Under 6 Months to 5 Years of Age
페이지 정보
작성자 VOA 뉴스 작성일 22-06-20 00:10 댓글 0본문
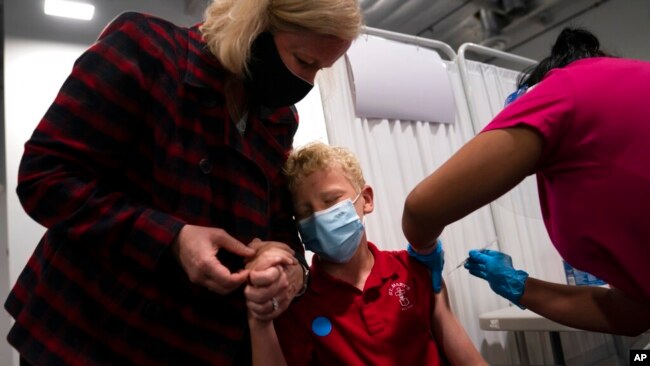
A 12-year-old boy in California, U.S., is being injected with a coronavirus vaccine in May last year. (Data picture)
On the 18th, the U.S. Centers for Disease Control and Prevention (CDC) approved vaccinations for infants from six months old to under the age of five.
This measure follows the unanimous recommendation of the CDC Advisory Committee on Vaccination (ACIP).
"We know that millions of parents and guardians are desperate to get their children vaccinated," CDC Director Rochelle Wallensky said in a statement. "With today's decision, we can now get vaccinated."
The decision will allow about 18 million infants and children to be vaccinated in the United States.
The vaccination is expected to begin next week.
Meanwhile, on the previous day (17th), the U.S. Food and Drug Administration (FDA) approved the vaccination of Pfizer and Moderna for infants from six months old to under five years old.
The Pfizer vaccine is inoculated three times and Moderna two times.
Pfizer says its vaccine is about 80 percent preventive.
Moderna announced that 51% of the preventive effects were confirmed between the ages of six months and two years and 37% between the ages of two and five.
VOA News
출처 : VOA한국
댓글목록 0
등록된 댓글이 없습니다.

















